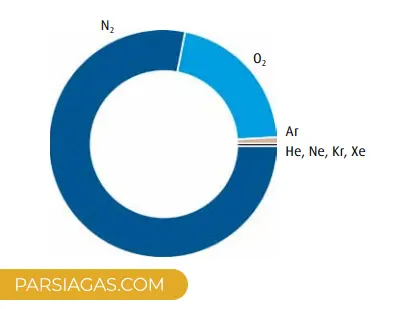
Air is one of the most valuable natural resources on Earth. While it may seem simple at first glance, it’s actually a complex and highly useful mixture of several key gases. Contrary to ancient beliefs that considered air a pure substance, we now know it consists of:
- Nitrogen (78%)
- Oxygen (21%)
- Argon (0.9%)
- Trace gases like helium, neon, krypton, and xenon
- Carbon dioxide and water vapor
These gases are essential not only for life on Earth but also in various industries such as healthcare, welding, laboratories, and energy production.
For this reason, Air Separation Technology is one of the foundations of modern industry.
In this article, we explore the most complete guide to methods of extracting gases from air.
What is Air Separation and Why Does It Matter?
Air separation is the process of isolating the main components of air—nitrogen, oxygen, and argon—in pure or liquefied form.
This process is vital in many sectors:
- Medical industry (medical oxygen)
- Steel production
- Industrial welding
- Food & beverage industry
- Oil & gas
- Chemical manufacturing
- Electronics
- Laboratory applications
In General, Air Separation Methods Are Divided into Two Main Categories:
1. Cryogenic Air Separation Methods
Gas extraction based on differences in boiling points
This technology is one of the oldest yet most advanced air separation methods.
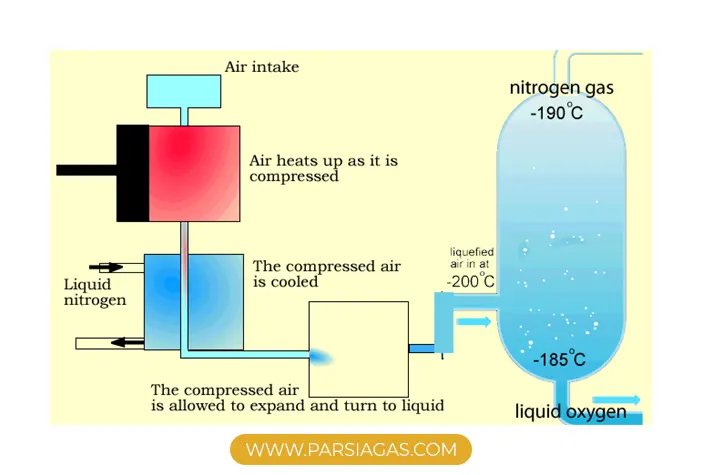
In this process, air is cooled to extremely low temperatures (approximately –180°C to –196°C) until its components liquefy. The gases are then separated through fractional distillation.
This technology was first developed by Carl von Linde in 1895 and later enhanced with modern advancements.
Complete Stages of Cryogenic Air Separation
Stage 1: Air Compression
Air enters multi-stage compressors, where it is compressed and pre-cooled.
Dust particles and mechanical impurities are removed at this stage.
Stage 2: Air Purification and Drying
Two major contaminants must be completely removed:
- Water vapor (H₂O)
- Carbon dioxide (CO₂)
These substances would freeze at cryogenic temperatures and block the system.
Common purification methods include:
- Adsorption using molecular sieves
- Reverse heat exchangers
Stage 3: Cryogenic Cooling
The purified air is cooled in counter-flow heat exchangers to temperatures close to its liquefaction point.
Stage 4: Fractional Distillation (Distillation Column)
The liquefied air enters a dual-column distillation system:
- Nitrogen exits from the top of the column (lower boiling point and density)
- Oxygen is collected from the bottom (higher boiling point)
- Argon is separated from the middle section of the column
To increase purity:
- A condenser is installed at the top
- A reboiler is located at the bottom
Stage 5: Product Compression
The separated gases—nitrogen, oxygen, and argon—are stored either:
- As compressed gases, or
- In liquid form (LIN / LOX / LAr)
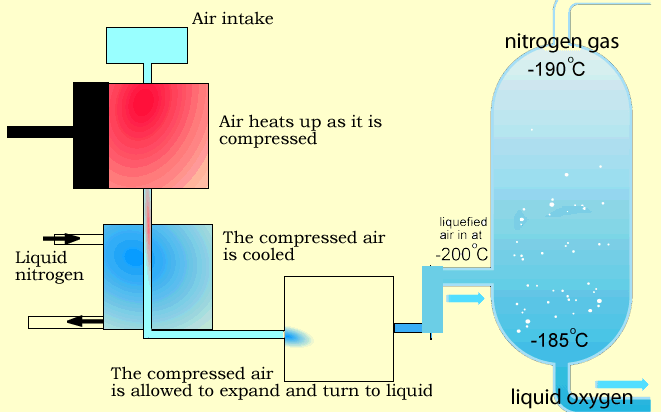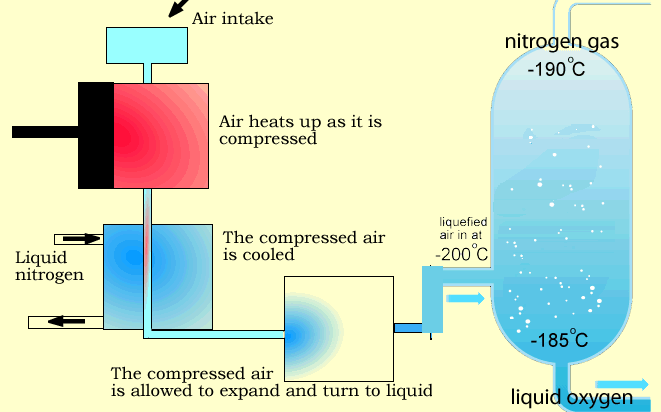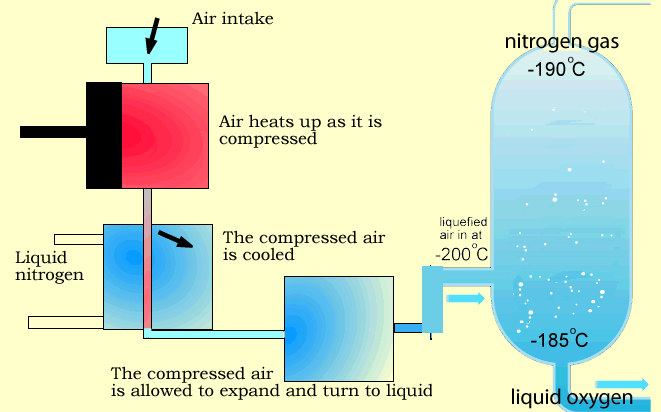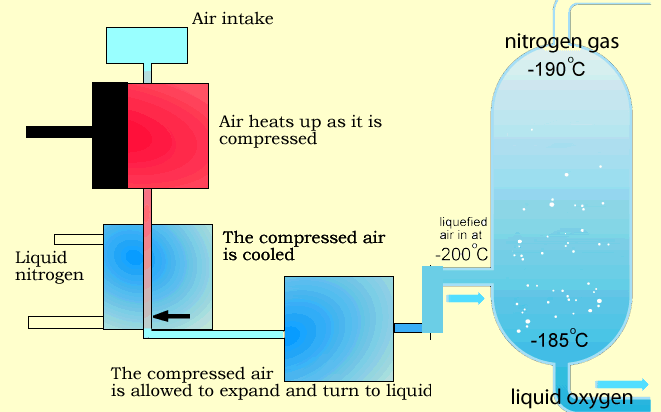
Why Is Liquid Product Generation Important?
For many industries, it’s essential to store gases in cryogenic liquid form, such as:
- Liquid Nitrogen (LIN)
- Liquid Oxygen (LOX)
- Liquid Argon (LAr)
Advantages of Liquid Storage:
- Reduced volume
- Easier transportation
- Delivery in industrial-scale quantities
- Quick conversion back to gas using vaporizers
2. Non-Cryogenic Air Separation Methods
These methods operate at ambient temperature and are cost-effective for medium-scale production of gaseous nitrogen and oxygen.
They include three main technologies:
2.1 Pressure Swing Adsorption (PSA) & Vacuum PSA (VPSA)
These methods use vessels filled with molecular sieves:
- In PSA, nitrogen is produced (oxygen is adsorbed).
- In VPSA, oxygen is produced (nitrogen is adsorbed).
Advantages:
- On-site gas generation
- Lower operational costs
- Adjustable purity (90%–99.9%)
2.2 Membrane Separation Technology
In this method, air passes through hollow fiber membranes.
- Fast-permeating gases (O₂, CO₂, H₂O)
→ exit quickly - Slow gases (N₂, Ar)
→ remain and are collected at the outlet.
Advantages:
- No moving parts
- Ideal for small and medium industries
- Suitable for 95%–99% nitrogen production
Detailed Comparison of Air Separation Methods
| Method | Purity | Scale | Output Form | Applications |
|---|---|---|---|---|
| Cryogenic | Very High (up to 99.999%) | Large Industrial | Gas + Liquid | Steel, Petrochemical, Medical, Welding |
| PSA / VPSA | Medium to High | Medium | Gas | Factories, Food Industry, Pharmaceuticals |
| Membrane | Medium | Small to Medium | Gas | Nitrogen in general industries |
Which Gases Can Only Be Extracted via Cryogenic Method?
- Argon
- Helium
- Rare gases (Neon, Krypton, Xenon)
No non-cryogenic method can extract these gases.
Why Is Air Separation Critical for Industry?
Because the three key gases from ambient air:
- Oxygen: For medical use, steel production, combustion, and chemical industries
- Nitrogen: Inert gas for food, electronics, welding, oil & gas
- Argon: Shielding gas for welding and laser technologies
…are all produced using these separation technologies.
A Note from ParsiaGas
With years of experience in supplying industrial, laboratory, and medical gases, ParsiaGas is capable of directly supplying:
- Nitrogen (gas and liquid)
- Oxygen (gas and liquid)
- Argon (gas and liquid)
- Rare gases
for a wide range of industries.
📞 For expert consultation and choosing the best gas supply method, contact ParsiaGas now.
Specialist Gas Consultation – Contact Us